Shorthand notation anode cathode1/12/2024 
In the cathode where reduction occurs, electrons must “jump” from the metal to a species in solution. It is also important to consider the portions of the cell where the metal interfaces with the solution. With these systems the conductivity is measured to determine the degree to which the water has been deionized (the reading is reported as a resistance and the higher the resistance, the less conductive the solution). One of the goals of these purification systems is to deionize the water. Many science buildings have a device that is designed to generate highly purified water. The conductivity of a solution directly correlates with the ionic strength of the solution. Conductivity is a measurement of the ability of a solution to conduct electricity. If a hairdryer is dropped into the water, the water conducts electricity because of ions in it with the end result that the person will be electrocuted. The ability of ions to conduct electricity is the reason why someone should never use a hairdryer while sitting in a bathtub full of water. In the solution, conduction of electricity is caused by migration of ions. For the metallic components (zinc, copper, copper connecting wire), electrons are responsible for the current flow. The processes responsible for the current flow in an electrochemical cell depend on which part of the cell you are in. 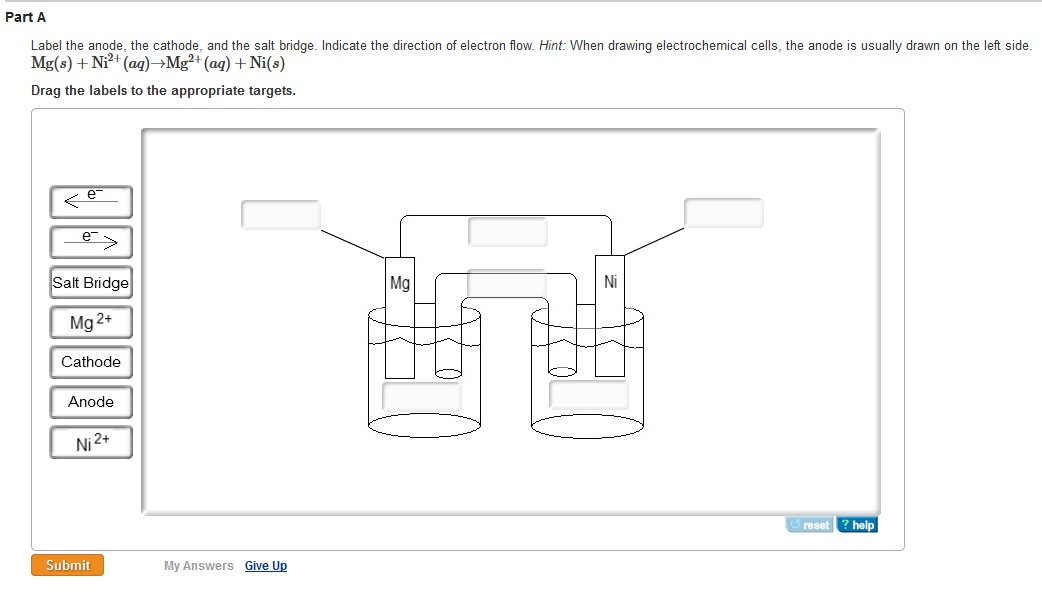
What processes are responsible for conduction of electricity in an electrochemical cell? The other is something known as a salt bridge that connects the two solutions. One is a metal wire that connects the two pieces of metal. Two connections are needed for a complete circuit. 
The two half-cells need to be connected to complete the circuitry and allow the reaction to proceed. If a half reaction does not form a solid metallic species (e.g., Fe 3 + + e – = Fe 2 +) an inert metal such as platinum is used in the cell. The copper half-cell consists of a piece of copper metal in a solution containing copper ion. The zinc half-cell consists of a piece of zinc metal in a solution containing zinc ion. By convention, the anode is always put on the left and the cathode on the right in the diagram. In an electrochemical cell, the reduction half reaction is referred to as the cathode and the oxidation half reaction is referred to as the anode. The salt bridge is represented by a double line, ‖.\]īased on the two E o values, the copper ion will be reduced and zinc metal will be oxidized. In this notation, information about the reaction at the anode appears on the left and information about the reaction at the cathode on the right. Electrochemical cells can be described using cell notation. The movement of these ions completes the circuit and keeps each half-cell electrically neutral. Anions in the salt bridge flow toward the anode and cations in the salt bridge flow toward the cathode. Adding a salt bridge completes the circuit allowing current to flow. The anode is connected to the cathode in the other half-cell, often shown on the right side in a figure. One half-cell, normally depicted on the left side in a figure, contains the anode. The half-cells separate the oxidation half-reaction from the reduction half-reaction and make it possible for current to flow through an external wire. These observations are consistent with (i) the oxidation of elemental copper to yield copper(II) ions, \ce(s) Check Your LearningĮlectrochemical cells typically consist of two half-cells. A copper wire and an aqueous solution of silver nitrate (left) are brought into contact (center) and a spontaneous transfer of electrons occurs, creating blue Cu2+(aq) and gray Ag(s) (right). A gradual but visually impressive change spontaneously occurs as the initially colorless solution becomes increasingly blue, and the initially smooth copper wire becomes covered with a porous gray solid.įigure 17.2.1. 
Use cell notation to symbolize the composition and construction of galvanic cellsĪs demonstration of spontaneous chemical change, Figure 17.2.1 shows the result of immersing a coiled wire of copper into an aqueous solution of silver nitrate.Describe the function of a galvanic cell and its components.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
